Posted: March 24, 2026

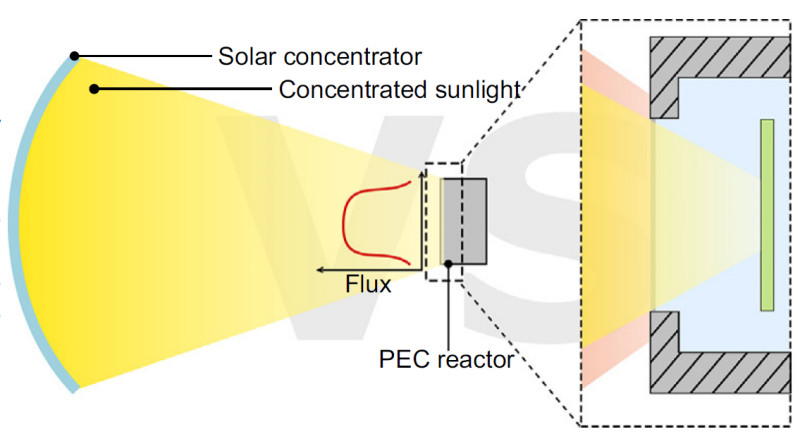
One of the strengths of the IEA Solar Heating and Cooling Programme is its ability to bring together scientists working on similar topics from across continents to exchange ideas. This was once again successfully demonstrated within Task 72 on Solar Photoreactors for the Production of Fuels and Chemicals. Prof. Craig McGregor from Stellenbosch University in South Africa attended the Task meeting in Portugal in October 2025 and was delighted by the gathering. McGregor is a dedicated concentrating solar power (CSP) expert with experience in both industry and academia, who has recently expanded his research portfolio to include green hydrogen technologies. His work follows a systems engineering approach, considering the photoelectrochemical reactor, the concentrator, and the balance of plant as a single integrated device.
You have just recently joined the Task 72 work. How were your experiences and lessons learned from participating in the last Task meeting in Portugal?
McGregor: It was a fantastic experience to join the workshop in Portugal. Fuel production with solar reactors is a new area of research for me, and it was great to meet and interact with experienced people in the field. If anything, the Task 72 meeting was too short; it would have been nice to get into some of the technology details, and have an opportunity to learn more.
As Associate Professor at the Centre for Renewable & Sustainable Energy Studies (CRSES) of Stellenbosch University your focus research field was CSP. Why did you start with green hydrogen technologies?
McGregor: Indeed, I started researching green hydrogen produced by water electrolysis a few years ago. These two disparate research areas come together in optical systems for photoelectrochemical processes. You need to get the photons to the electrochemical reactor for this technology to work. This was an opportunity to leverage my CSP experience to improve the reactor technology.

Demonstrator with a 60 cm2 photoelectrochemical reactor for hydrogen production tested under outdoor conditions
Photo: Stellenbosch University
In a recently published paper [1] you followed a system engineering approach analysing the interfaces between three components of a photoelectrochemical (PEC) solar-fuel systems: photoelectrochemical reactor, concentrators and balance of plant. Why is the advantage of this system approach?
McGregor: In our field, there is a natural tendency to work in "silos." Materials scientists focus on semiconductors, while mechanical engineers like myself focus on optics. The problem is that when you try to bolt these two together in the real world, you run into interface issues that haven't been accounted for.
The advantage of a systems engineering approach is that it forces us to look at the system of interest as a whole from day one. Instead of just asking, "How do we make a better concentrator?", we ask, "What exactly does the PEC reactor need from the concentrator to thrive?".
By identifying these requirements early, we realised that standard silvered-glass mirrors actually reduce the specific short-wavelength light - the UV and visible spectrum - that many photoelectrodes need to drive the reaction. We also addressed the critical trade-off between optical efficiency and flux uniformity; because researchers are so worried about hot spots, they tend to overvalue flux uniformity without realising the severe penalty it imposes on the solar concentrator's efficiency. Finally, this approach integrates the balance of plant to ensure that as input energy fluctuates, the system can actively manage thermal loads and bubble removal to maintain stability. Ultimately, this shifts the field away from trial-and-error toward a structured framework to develop a competitive cost of hydrogen.
Experimental setup for a prototype photoelectrochemical reactor with concentrating optics
Graphic:Stellenbosch University
In your paper you conclude that concentrator optics must be tailored to chemistry not vice versa, why?
McGregor: In my background field of CSP, the whole goal of a concentrator is to create as much heat as possible often using concentration ratios over 2,500 - to maximise turbine efficiency. But PEC chemistry simply can't survive that. Photoelectrodes are highly sensitive to temperature; for example, materials such as BiVO4 - Bismuth Vanadate - begin degrading at temperatures just above 40 °C. The elevated heat accelerates side reactions and photocorrosion. Therefore, we can't just bolt a standard CSP concentrator onto a PEC reactor. We have to specifically tailor the optics to provide a 'sweet spot' with moderate concentration, typically around 10 to 50 suns, so we boost the chemical production rate without cooking the reactor. And we have to do this with reflective materials with the right optic (bandgap) properties.
Why is it important to consider the balance of plant (BoP) which includes pumps, cooling, storage, control, safety, etc. when developing a pilot electrochemical reactor plant development?
McGregor: Let's look at this from a very practical, physical perspective. To get enough sunlight, our solar concentrators have to physically move to track the sun throughout the day. But inside the reactor, we are splitting water and creating bubbles of hydrogen and oxygen gas. Naturally, these bubbles want to rise straight up due to gravity. So, if your reactor is constantly tilting to follow the sun, how do you reliably get the bubbles out?
This is exactly where the BoP becomes a critical piece of the core technology, rather than just basic plumbing. One elegant solution is to use the BoP's pumping system to push the liquid electrolyte through the reactor fast enough that the fluid's shear force simply sweeps the bubbles away, completely overriding the effects of gravity. As a fantastic bonus, pushing all this fluid through the system also dissipates the excess heat generated by concentrated sunlight, effectively solving our thermal management problem at the same time.
However, as with all engineering, there is a catch. We can't just install a massive pump and blast fluid through the system. First, running a large pump causes parasitic energy losses that erode the plant's overall efficiency and economics. Second, if the fluid moves too violently, the shear stress can erode and mechanically degrade the delicate photoelectrodes.
So, developing the BoP is really about striking the perfect dynamic balance: pumping just enough fluid to keep the reactor cool and free of bubbles, without damaging the materials or undermining the plant's economics.
What are the conclusions from the paper for your pilot electrochemical reactor plant development of your research group?
McGregor: The main takeaway for our pilot development is that we must design for survival in unpredictable outdoor conditions rather than peak efficiency in a steady-state laboratory. For instance, our recent field tests revealed that passing clouds don't just temporarily reduce output; they also create severe voltage mismatches between the photovoltaic and photoanode components, which actively degrade the semiconductor materials. Our next pilot iteration must address this.
We also learned that we cannot overlook practical outdoor realities, such as our electrolyte crystallising in storage tanks during overnight temperature drops. Finally, we are focusing our future optical designs on simplified, one-dimensional solar tracking. Our systems analysis shows this approach provides the necessary concentration and flux uniformity to protect the reactor while keeping capital costs realistic for commercial scale-up.
[1] Arend Wessels Moelich, Craig McGregor: A systems engineering review on the integration of solar concentrators with photoelectrochemical processes, Elsevier 2026, https://doi.org/10.1016/j.rser.2026.116792
Websites of organisations mentioned in this news article: